Other naturally occurring hydrocarbons with double bonds include plant pigments such as lycopene, which is responsible for the red colour of ripe tomatoes and watermelon. α-Pinene is used as a paint thinner as well as a starting material for the preparation of synthetic camphor, drugs, and other chemicals. Oil of turpentine, obtained by distilling the exudate from pine trees, is a mixture of hydrocarbons rich in α- pinene. (Essential oils are responsible for the characteristic odour, or “essence,” of the plant from which they are obtained.) Myrcene and limonene, for example, are alkenes found in bayberry and lime oil, respectively. The biosynthesis of ethylene involves an enzyme-catalyzed decomposition of a novel amino acid, and, once formed, ethylene stimulates the ripening of fruits.Īlkenes are abundant in the essential oils of trees and other plants. Natural occurrenceĮthylene is formed in small amounts as a plant hormone. Cyclooctyne (C 8 H 12) is the smallest cycloalkyne capable of being isolated and stored as a stable compound. trans-Cycloalkenes are too unstable to isolate when the ring has seven or fewer carbons.īecause the C―C≡C―C unit of an alkyne is linear, cycloalkynes are possible only when the number of carbon atoms in the ring is large enough to confer the flexibility necessary to accommodate this geometry. (An alternative method, based on the Cahn-Ingold-Prelog system and using E and Z prefixes, is also used.) Cycloalkenes in which the ring has eight or more carbons are capable of existing as cis or trans stereoisomers. In most cases, the names of stereoisomeric alkenes are distinguished by cis- trans notation. 
Stereoisomerism is therefore possible in those alkenes in which neither carbon atom bears two identical substituents. Unlike rotation about carbon-carbon single bonds, which is exceedingly rapid, rotation about carbon-carbon double bonds does not occur under normal circumstances. In naming substituted derivatives of cycloalkenes, numbering begins at and continues through the double bond. Dienes are classified as cumulated, conjugated, or isolated according to whether the double bonds constitute a C=C=C unit, a C=C―C=C unit, or a C=C―(CXY) n―C=C unit, respectively.ĭouble bonds can be incorporated into rings of all sizes, resulting in cycloalkenes. Dienes are named by replacing the -ane suffix of the corresponding alkane by -adiene and identifying the positions of the double bonds by numerical locants. 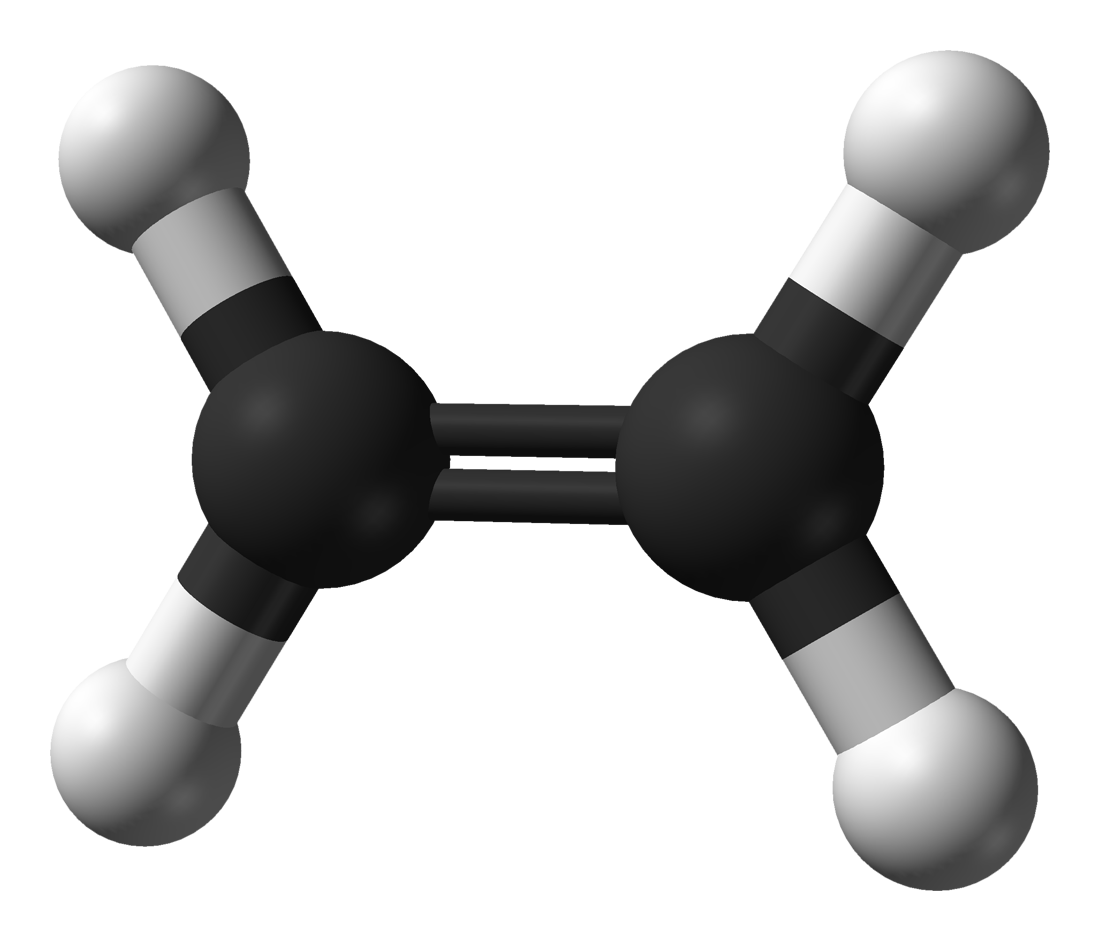
Once the chain is numbered with respect to the multiple bond, substituents attached to the parent chain are listed in alphabetical order and their positions identified by number.Ĭompounds that contain two double bonds are classified as dienes, those with three as trienes, and so forth. The chain is numbered in the direction that gives the lowest number to the first multiply bonded carbon, and adding it as a prefix to the name. Higher alkenes and alkynes are named by counting the number of carbons in the longest continuous chain that includes the double or triple bond and appending an -ene (alkene) or -yne (alkyne) suffix to the stem name of the unbranched alkane having that number of carbons. SpaceNext50 Britannica presents SpaceNext50, From the race to the Moon to space stewardship, we explore a wide range of subjects that feed our curiosity about space!Įthylene and acetylene are synonyms in the IUPAC nomenclature system for ethene and ethyne, respectively.Learn about the major environmental problems facing our planet and what can be done about them! Saving Earth Britannica Presents Earth’s To-Do List for the 21st Century.Britannica Beyond We’ve created a new place where questions are at the center of learning.100 Women Britannica celebrates the centennial of the Nineteenth Amendment, highlighting suffragists and history-making politicians.COVID-19 Portal While this global health crisis continues to evolve, it can be useful to look to past pandemics to better understand how to respond today.Student Portal Britannica is the ultimate student resource for key school subjects like history, government, literature, and more.This Time in History In these videos, find out what happened this month (or any month!) in history.#WTFact Videos In #WTFact Britannica shares some of the most bizarre facts we can find.Demystified Videos In Demystified, Britannica has all the answers to your burning questions.Britannica Explains In these videos, Britannica explains a variety of topics and answers frequently asked questions.Britannica Classics Check out these retro videos from Encyclopedia Britannica’s archives.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
